menu
PRIMS is a complete compliance and safety software solution for cosmetics brands, (3rd party) manufacturers, and consultants.
The cloud-based platform is available 24/7 and provides real-time visibility of portfolio safety and compliance based on the latest regulatory and toxicological data. Teams work from a single, consistent source of information to manage compliance requirements across the EU, UK, USA (MoCRA), and Canada, including retailer-specific restrictions.
PRIMS is developed and continuously updated by The Regulatory Company’s Safety Assessors and Regulatory Specialists.
Specifications management
PRIMS structures raw materials, formulas, packaging components, and finished products within one connected system. Each specification is managed with its own versioning, lifecycle status, and full traceability down to the INCI level. Cloning tools allows to quickly create product updates or variations without rebuilding specifications from scratch.
The PIF completeness view highlights missing documentation so the PIFs are ready for audits and market launch.
PRIMS can also connect to your existing systems through API integrations.
Regulatory and toxicology data, smart algorithms
PRIMS integrates regulatory and toxicological data at substance level (INCI), continuously maintained by the in-house experts at The Regulatory Company. Safety information is sourced from recognised authorities such as CIR, EFSA, WHO and OECD and supported by evidence-based risk assessment methods. When toxicological data is incomplete, PRIMS supports NGRA approaches including TTC and read-across. Regulatory updates and amendments are added as they become available, ensuring that compliance across your product portfolio is monitored in real time.
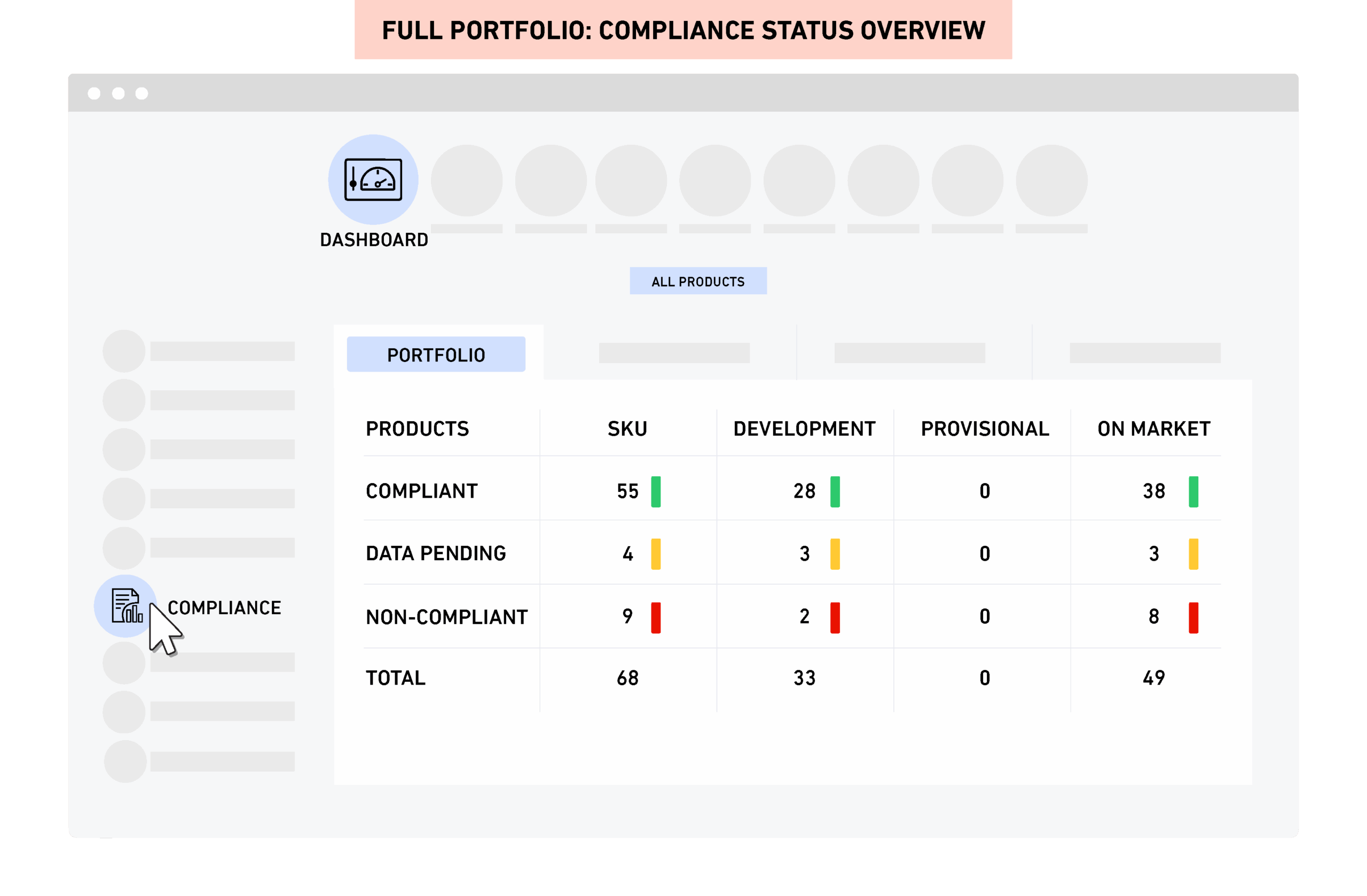
Dashboards and reporting
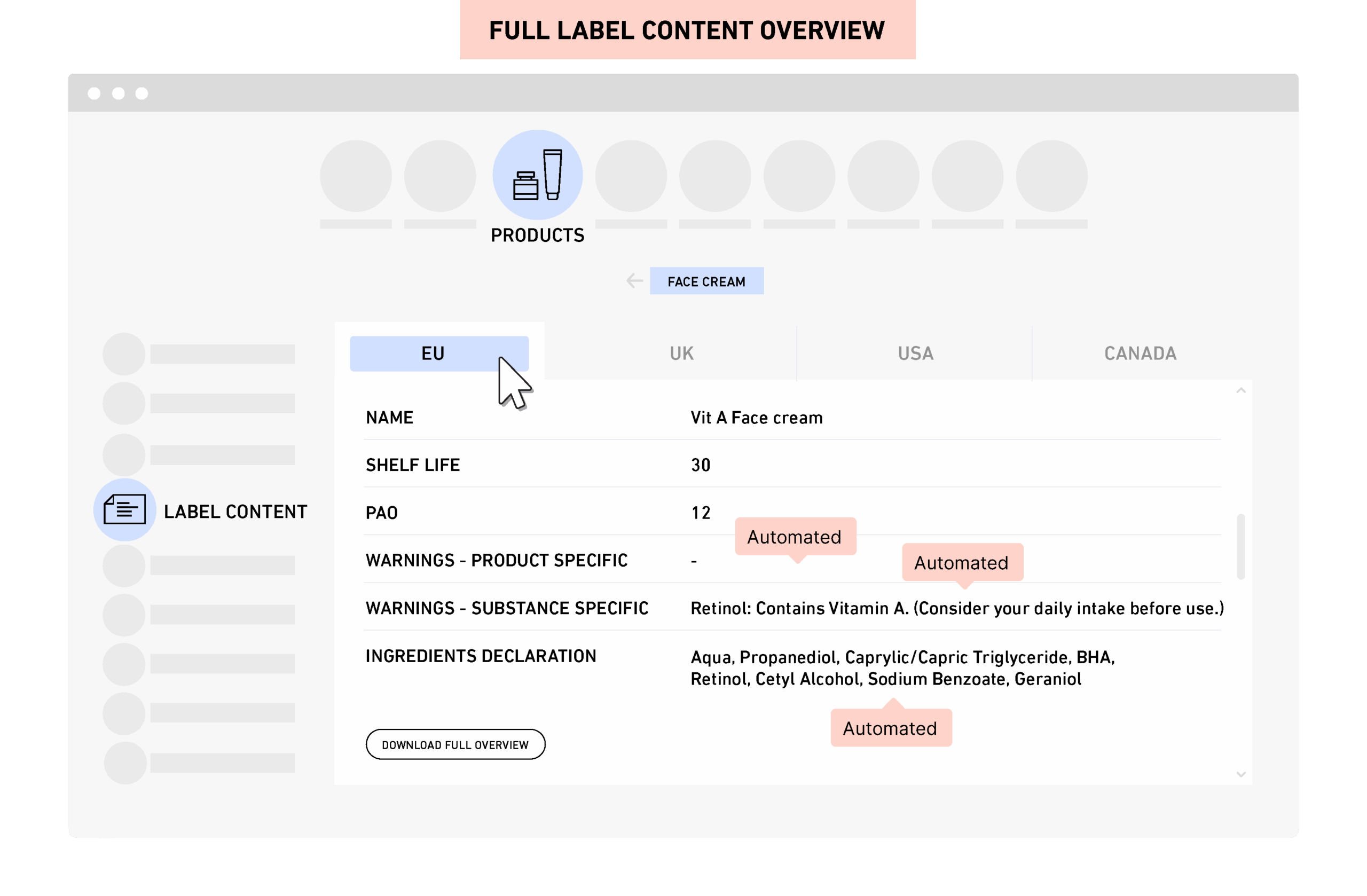
PRIMS gives real time compliance and safety visibility across the full portfolio. You can access product specific analysis with one click and generate label content and cumulative compositions instantly.
Safety Assessmens can be quickly drafted, based on exposure scenarios and toxicology thresholds ready for review and approval.
The REACH dashboard calculates imported volumes and thresholds per substance and supplier.
"Where-used" views show which products or materials are impacted by regulatory updates.



Contact us and have your portfolio pre-assessed for free! We will look into your needs, your product formulations, your product label and marketing materials, and discuss your product dossiers.
We fully support the data transfer to ensure a smooth transition, a minimum-effort implementation process, and short-term internal adoption of PRIMS.
You provide the compositional data and other structured data in bulk. We perform the data import and we make sure that your PRIMS users are properly trained. You are guided in the entire on-boarding process by one of our dedicated project managers that will be assigned to your project.
Already on the first access to PRIMS most features and reporting are immediately available, such as automated compliance status monitoring and detailed analysis per product, product composition downloads and mandatory label content briefs, 24/7 real-time compliance dashboard (including the REACH management dashboard), and where used reporting.
Fast
Import raw material and formulation data in bulk and manage specifications efficiently with intuitive versioning and cloning tools. PRIMS performs automated compliance and safety analysis and generates mandatory label content, PIF documentation, and registration files in one click. REACH reports can also be created instantly.
Less resources
PRIMS follows a “one specification, one place” principle. All specifications are connected and automatically synchronised, allowing updates to flow through the entire portfolio. Impact analysis, reporting tools, and compliance dashboards provide clear visibility of product status and PIF completeness, while PIFs, CPSRs, and label content can be generated and exported in real time.
High quality data
Regulatory and toxicological databases are included and maintained by The Regulatory Company. They are continuously updated with the latest regulatory amendments and scientific data, ensuring that product safety and compliance status are always current. Historical insights and portfolio-wide compliance visibility are available at any time.
Reliable
PRIMS is already used by customers and TRC to manage thousands of cosmetic products. The system provides full transparency across the entire product structure, from substance level to finished product. Users also benefit from the support of TRC’s regulatory consultancy, compliance services, and Responsible Person expertise, with a strong track record of successful competent authority inspections.

PRIMS includes an intuitive user interface, dashboard overview with product portfolio compliance status and easy navigation. Specifications are fully traceable back and forth through one-click navigation between raw materials, formulations, packaging components, products and companies (suppliers), and where-used reports are generated and exported in spreadsheet format in a few seconds.

PRIMS has been developed with a 100% focus on regulatory requirements and processes, and to quickly produce the relevant output documents and reports such as the CPSR, Cumulative Product Composition (exact or ranges), and the Regulatory Content Brief (mandatory label content).
Documents and reports are available in PRIMS at all times, are quickly (re-) generated to adjust for any changes, and ready for export/download to all users.
The REACH management dashboard presents a real-time overview of all substance-supplier combinations that require verification of REACH registration compliance.
To manage importer obligations, PRIMS algorithms calculate and present substance-supplier combinations where the annually imported quantity surpasses the 1.000 kg threshold, by at least one individual importer, and where the substance is not exempted.
By changing the selection criteria (calendar year, region, local manufacturers), the dashboard overview is modified to meet your needs.
PRIMS enables the regulatory team to work faster and more efficiently, and adds significant quality, continuity and confidence to the regulatory function. The compliance and safety results are highly reliable.
Once operational (within 4-8 weeks), total resources spent on regulatory processes are significantly reduced with up to 40%, in particular on PIF management, and (continuous) compliance and safety assessment.
